Posts in Category: projects
Coccinella
What is coccinella?
Coccinella is an innovative open-source framework developed for high-throughput behavioral analysis. Leveraging the power of distributed microcomputers, it facilitates real-time tracking of small animals, such as Drosophila melanogaster. Complementing this tracking capability, coccinella employs advanced statistical learning techniques to decipher and categorize observed behaviors. Unlike many high-resolution systems that often require significant resources and may compromise on throughput, coccinella strikes a balance, offering both precision and efficiency. Built upon the foundation of ethoscopes, this platform extracts minimalist yet crucial information from behavioral paradigms. Notably, in comparative studies, coccinella has demonstrated superior performance in recognizing pharmacobehavioral patterns, achieving this at a fraction of the cost of other state-of-the-art systems. This framework promises to complement current ethomics tools by providing a cost-effective, efficient, and precise tool for behavioral research. Coccinella analysis can be done in ethoscopy, a Python framework for analysis of ethoscope data.
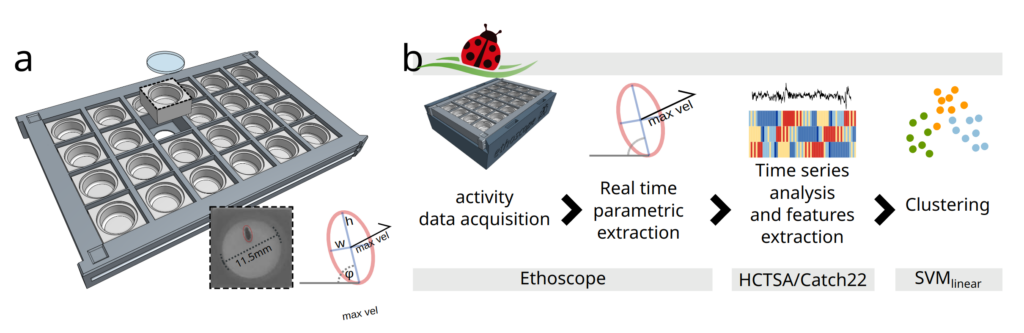
How does it work?
Coccinella uses ethoscopes to extract information about the activity of flies in real time. Ethoscopes are machines that use distributed computing via Raspberry PI to detect and interfere with behaviour. Given the off-the-shelf nature of the devices, the all setup is inexpensive and scales up very easily. As term of reference: our lab currently employs about 100 ethoscopes, with a processing power of 20 flies each.

Data about the activity of the animal are then fed to a high-throughput toolbox for time series analysis called HCTSA or Catch22, initially developed at Imperial College London by our colleagues in the Maths Department. The toolbox performs numerous statistical tests aimed at segregating data in an unsupervised way and can therefore be used to cluster together data that the machine recognises as similar. In our case, we tried to identify which drugs have similar mode of action, potentially recognising and assigning the appropriate pharmacological pathways to new, uncharacterized compounds.
We also compared the performance of coccinella to state of the art systems and found that it performs even better!
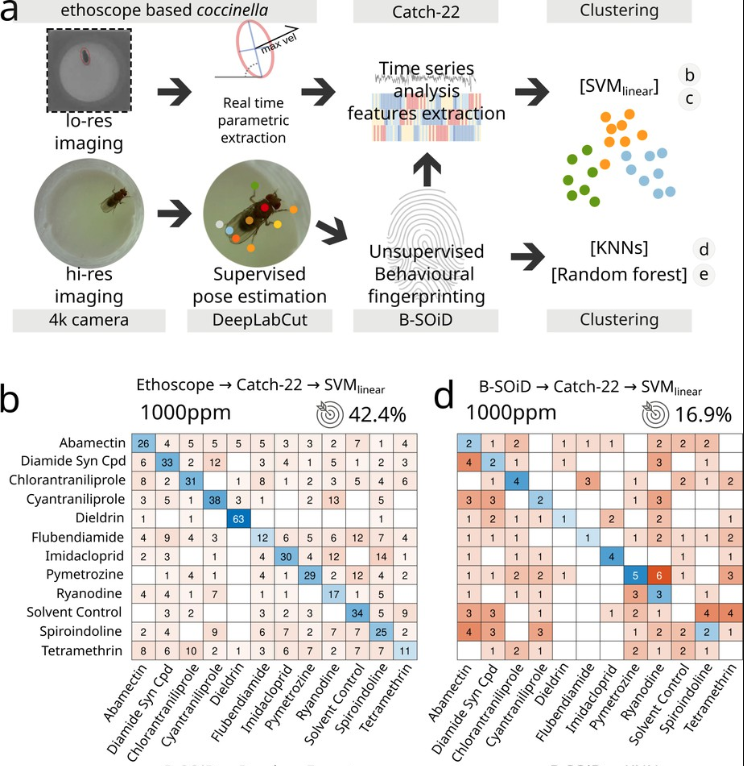
This is important from the technical point of view but also from the standpoint of neuroscience because it shows that “less is more” when it comes to extracting and recognising behavioural data. In other words, you don’t need to carefully label posture and movement when characterising behaviour: reducing activity to its minimal terms actually works even better!
 | Coccinella paper on eLife |
 | Ethoscopy / Ethoscope-lab paper on Bioinformatics Advances |
 | Ethoscopy on GitHub |
 | Ethoscopy on PyPi |
| Ethoscope-lab Docker container on DockerHub | |
 | Jupyter Notebook tutorials for Ethoscopy on GitHub |
 | Ethoscopy and Ethoscope-lab documentation on bookstack |
| Raw data and all notebooks reproducing the paper’s figures on Zenodo |
Ethoscopy and Ethoscope-lab
- Ethoscopy is Python software for analysis of ethoscope data – and more! – created by Laurence Blackhurst
- Ethoscope-lab is a pre-baked Docker container featuring an installation of the multi user Jupyter Hub with Python and R kernels, ready to be used with Ethoscopy and Rethomics.
Why using ethoscope-lab?
Let me simply explain how we use it in our lab. We arranged a powerful workstation that acts as lab server and run a dockerized ethoscope-lab on it. The workstation has a local copy of all our ethoscope data (about 8 Terabyte as I type) and ethoscope-lab has local access on those, offering the quickest loading time. Users can then use their computer, or tablet to connect to the workstation and perform data analysis directly from the browser. The setup frees them from working at their desk and allows access to their data from anywhere in the world, guaranteeing at the same time the fastest computational performance even when they work on their laptops. Moreover, the system uses Jupyter notebook as default, meaning each analysis can be nicely annotated and exported to be shared with the world post-publication, along the original raw data.
To give a practical example: this series of repositories on zenodo contains the entire dataset of our latest paper (316Gb) and it’s paired to all the notebooks we used to generate each figure. Readers can download the dataset freely, install ethoscope-lab as docker container on any computer (irrespective of the operating system they adopt) and reproduce all our analyses!
Understanding the links between sleep and well being – lessons from fruit flies
Drosophila melanogaster, commonly known as fruit fly or more appropriately vinegar fly, is the second most common animal model used in research. Initially established by Thomas Hunt Morgan at the beginning of 1900s to provide an empirical base to the groundbreaking hypotheses of Darwin, flies have contributed to science in every realm, from development to genetics and neuroscience. 6 Nobel prizes have been awarded to flies in the past century, with the last one being awarded in 2018 to three Drosophila researchers for their work in the characterisation of circadian behaviour.
My laboratory uses flies to try and answer one simple, yet fascinating question: why do we sleep? What is sleep for and why humans and all animals seem to require sleep? Our approach is somehow different mainstream one, because flies force us to think to the problem in an unusual and more creative way. So far this has paid off egregiously, and we have managed to make very important discoveries that apply well beyond the fly realm.
One aspect of our research that may be particularly interesting for someone puzzled about the science of physical well being, is that we do not consider sleep to be a prerogative of the brain but actually a phenomenon that affects every part of our body. Whenever we lack sleep – whether for fun or for work – we can feel an effect of sleep deprivation not just on our cognitive performance but on our very body too. This is more than a subjective feeling: it is a well-established phenomenon in humans and animals. Why is it so? Studying sleep deprivation in flies we hope to give an answer to this question.
In particular, we use state of the art technology to a) deprive flies of sleep employing custom-made robots and b) exploring what changes at the cellular level when we lack sleep. How genes, proteins, and other molecules change their composition when we lack sleep?
We can use the same robotic technology to also study physical behaviour in these animals: are they in good shape? Can they climb a wall as they normally do? Can they fly with the same stamina and precision? A great amount of literature in the field of muscular degeneration has been obtained in fruit flies and we have learned a great deal about genes controlling these aspects and how they fail in disease.
A very important corollary of our research is that understanding the functions of sleep opens the door to what we somehow half-jokingly call “the sleep pill”. If could understand what aspects of sleep make us refreshed and performing – both behaviourally and physically – we could then replace sleep pharmacologically. Or we could consolidate the beneficial aspects of sleep to increase its restorative power. To do that, we first need to understand what sleep is and what it does.
What would we do with a philanthropic donation?
We would employ the money to retain a brilliant young research assistant for a year or longer so that she could continue working on her project. The student recently graduated from an MSci in Neuroscience and she joined our laboratory for a summer placement in order to gain first-hand insights into our research. If she could stay longer, she could join and potentiate the research line of the laboratory that looks at the direct consequences of sleep deprivation. I have been working with Imperial Alumni who were kind enough to donate to my laboratory in the past. It has been a great honour and a pleasure and I am looking forward to doing this again.
Automatic detection and analysis of animal behaviour
Description
Even simple animals, like fruitflies or worms, can make complex decisions, for instance when they interact with predators, possible sex mates or explore food sources. Ethomics is a new discipline of neuroscience, attempting automatic and high-throughput analysis of animal behaviour, and investigating correlations and links to explore how genes drive behaviour. The purpose of this project is to create state of the art techniques of computer vision and machine learning to track animals activity and link them to stereotypical behaviours. Ultimately, we aim at building a system that can recognize animals status or intention and interfere with them using for instance laser or mechanical stimulations.
In particular, an initial application of the machine will be to create a new paradigm to study the links between sleep and learning in Drosophila. This new tool will be used for automatized sleep deprivation and learning conditioning. In the past, we created a software to track Drosophila locomotor activity and analyse sleep. The system, released as open source, is highly scalable, offers high resolution, it is affordable and easy to use (www.pysolo.net). With this project, we aim at expanding the existing system so that it would no longer be limited to passive detection of flies activity, but could actively interact with a group of animals, shining a heat-transducing infrared laser beam onto single flies whenever specific conditions are satisfied.
Project timeline.
Data analysis, computer vision, machine learning, component.
Techniques used: mathematics and statistics applied to data analysis and computer science
- The existing software is already able to track the animals position in real time and it would need to use this information to direct one or more IR laser diodes connected to motors. However, it will be necessary to modify the video tracking to recognize multiple interacting flies with higher precision and target only those that satisfy predefined conditions . This part will be done in collaboration with Dr. Stefanos Zafeiriou of the computing department at Imperial College.
- In particular, the software will be modified so that “conditions for shooting” will easily be programmed by the researcher. Also, the software should be able to recognize and output a complete data frame regarding the previous, current and predicted conditions of each flies. This will achieved using machine learning algorithms to identify stereotypic behaviours and act accordingly.
Building and testing the hardware component.
Techniques used: principle of electronics, basic physics of lasers
- Once flies are successfully tracked and conditions established, the software will interact with a motor controller to aim and shoot one or more laser diodes onto single flies
- Laser pulses will have to be theoretically selected and empirically calibrated for wavelength, power and duration, to use them as sleep deprivators or for learning conditioning
- Final protocols will be established for the different possible uses of the machine that will combine part 1 and part 2 of the project
Additional material and links.
- Perona’s and Dickinson’s software for automated fly behaviour is found at this address and described in this paper.
- Another software, again from Perona, is described here.
- A software for ant tracking can be found here, and here are some samples of how it performs.
- A sample of my flies moving in a vial (~15 minutes video at VGA resolution) can be found here
- Other sample movies from the ctrax project can be found here and here
Funding.
This project is funded by a Royal Society research grant.
Ethanol and sleep
Background
Ethanol is an evolutionary conserved neuromodulating agent, effective in mammals as it is in invertebrates. The fruitfly Drosophila melanogaster responds to ethanol with all the stereotypical signs that are also observed in humans: including euphoria, sedation, habituation and addiction (for a review see 1). Genetic predisposition in humans is accounted to be responsible for about 50% of the risk of developing addiction to ethanol, a major medical and social problem in modern society. For all these reasons, Drosophila has successfully been used in the past decades to investigate the genetic and molecular components of ethanol effects in the brain.
Hypothesis Student will Investigate
The student will investigate how ethanol affects the sleep / wake cycle of Drosophila and, conversely, how the sleep / wake cycle affects the behavioural and molecular responses to ethanol. Some questions that the student will address are: is the sedation induced by high concentrations of ethanol similar to sleep, with all the restorative effects associated to it? What are the effects on the sleep / wake cycle of chronic consumption of ethanol? In flies, Response to ethanol has been shown to be under partial control of the genes regulating the circadian clock and regulating synaptic output in the brain[2], and the same is true for sleep[3]: what is the biological relevance of this observation.
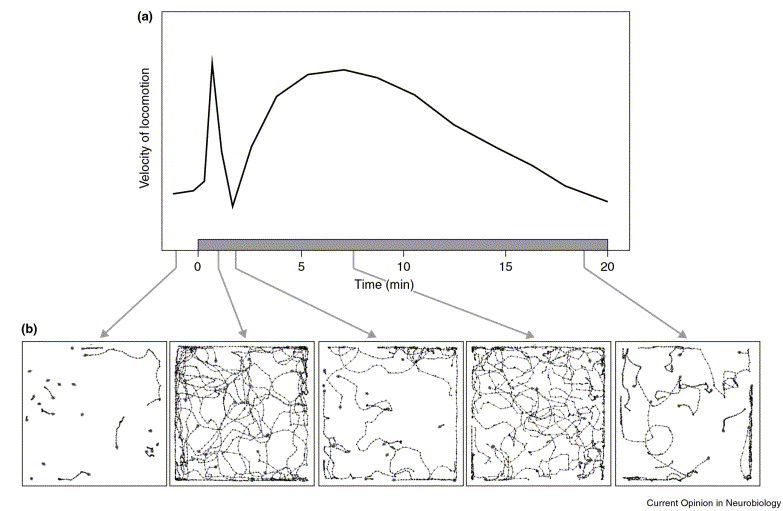
Effects of ethanol on Drosophila locomotion (a) Representation of the locomotor velocity of wild-type flies during an exposure to a moderate dose of ethanol (ethanol exposure period is shown by the grey horizontal bar). (b) Computer-generated traces of the locomotor behavior of a group of 20 flies before and during exposure to ethanol vapor. Each panel corresponds to a 10-s time period recorded at the times indicated in (a). Reproduced from (1)
Techniques Student will Use
The student will perform behavioural experiments to investigate the physiological responses to ethanol sub ministration: this will include assaying sleep, anesthesia, sedation, motility as well as learning and memory by mean of Drosophila learning paradigms. The student will also perform anatomical dissections and molecular analysis exploring how gene expression changes in the brain upon ethanol administration.
References and recommended readings
- Drugs, flies and videotape: the effects of ethanol and cocaine on Drosophila locomotion.
Curr Opin Neurobiol. 2002 Dec;12(6):639-45. (pdf) - arouser reveals a role for synapse number in the regulation of ethanol sensitivity.
Neuron. 2011 Jun 9;70(5):979-90. (pdf) - Widespread changes in synaptic markers as a function of sleep and wakefulness in Drosophila.
Science. 2009 Apr 3;324(5923):109-12. (pdf)
Sleep and learning: a genetic approach. The allnighter gene.
Background
Sleep is a vital activity, whose function still remain mysterious despite centuries of scientific research. All animals that have been tested so far, from nematodes to humans, possess and require the fundamental characteristics of sleep. In Drosophila, like in humans, sleep deprivation leads to a remarkable decrease in intellectual performance, learning and memory; chronic sleep restrictions also shows widespread metabolic changes and eventually leads to unexplained death.
My laboratory investigates the many functions of sleep using mainly the fruit fly Drosophila melanogaster as model organism. In particular, current research is aimed at elucidating the connections between sleep and synaptic plasticity, learning and neuronal homoeostasis. In previous work we provided evidence of how sleep may function as a mechanism to maintain a proper homoeostasis for synaptic strength and connections in Drosophila (see [1] for a recent review). We are now extending that line of work and we employ a rich selection of multidisciplinary techniques ranging from genetic manipulation of Drosophila (with transgenes and RNAi) to computer assisted analysis of behaviour to measure intellectual performance, including odours recognition and ability to court and mate.
The project
Following a genome wide screening for short sleeping mutant flies, we identified a novel gene that we called allnighter. allnighter mutant flies are viable but sleep considerably less than wild type controls and show general symptoms (such as tense “eagle” wings) that strongly suggest an underlying problem with neuronal excitability.
The project aims at extending the characterization of this gene, and other belonging to the same family. Some of the questions that you will try to answer are: “where is the gene express and at what stages of development? Does expression change with sleep or experience? Is the enzymatic activity of allnighter required for its function? How do allnighter flies perform when challenged with task measuring their learning and memory capabilities”
Techniques. Working with Drosophila
An MRes rotation in Drosophila allows the unique opportunity to investigate a biological problem in vivo and yet follow the development of a relatively complicated project from the beginning (e.g.:genetic manipulation of a new animal) to the end (e.g.: behavioural testing of the new phenotype). Your daily work will most likely encompass basic techniques of molecular biology (DNA cloning, PCR etc), genetics (crossing flies and follow up progeny) and behavioural neuroscience (analysis of sleep, sleep deprivation, analysis of learning and memory performance).
Drosophila and neurobiology.
For decades, Drosophila has been the most powerful animal models for genetic dissection and manipulation, and the outstanding contributions that flies gave to developmental biology and genetics were celebrated twice with Nobel Prizes(1933, 1995), and countless time in our text books. Recently, more and more laboratories started pairing the incredible genetic tools that we have been building in the past century with new and exciting neuronal techniques, leading to a Drosophila neuronal renaissance. From circuit formation to their function, Drosophila offers the complexity of an animal that can learn, memorize, socialize and yet the accessibility of a 250 thousand neurons brain. Here, I link a few entertaining and informative videos with the aim to communicate the excitement this field is living right now.
- Gero Miesenboeck (Oxford). Engineering the brain (18 minutes TED talk)
- Michael Dickinson (Caltech). Towards an integrated view of brain function (28 minutes video)
- Bjoern Brembs (Berlin). The Drosophila Flight Simulator (3 minutes video)
- Charalambos Kyriacou (Leicester). An interview on the use of Drosophila for studying circadian biology and behaviour (14 minutes video)
Getting in touch.
My office is in room 743 of the Huxley Building, in the South Kensington Campus.
Email and phone number are listed here. I’ll be happy to meet you and show you the lab, just drop me an email.
References and sample readings
- Synaptic plasticity in sleep: learning, homeostasis and disease.
Trends Neurosci. 2011 Sep;34(9):452-63. (pdf) - Waking experience affects sleep need in Drosophila.
Science. 2006 Sep 22;313(5794):1775-81. (pdf) - Widespread changes in synaptic markers as a function of sleep and wakefulness in Drosophila.
Science. 2009 Apr 3;324(5923):109-12. (pdf)
